-
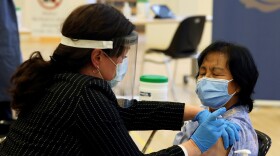
The Biden administration says it plans to buy an additional 100 million doses from both Moderna and Pfizer and pledges greater transparency with state and local leaders about supply expectations.
-
Leaders from across the European Union's 27 member-states celebrated the start of vaccination efforts on Sunday. EU Commission President Ursula von der Leyen called it "a European success story."
-
"Scientifically, it is highly likely that the immune response by this vaccine also can deal with the new virus variant," BioNTech CEO Ugur Sahin said Tuesday.
-
Hours after the EU's drug agency signed off on the vaccine, the European Commission approved its use, clearing the way for inoculations to begin across Europe.
-
The Food and Drug Administration greenlights the biotech firm's vaccine for emergency use in the U.S. The move bolsters a vast inoculation effort that's already underway.
-
Scientists and investors born outside the U.S. played crucial roles in the development of COVID-19 vaccines — a remarkable vindication for the argument that innovation depends on immigration.
-
Two days before a panel of experts is set to review Moderna's COVID-19 vaccine and advise the Food and Drug Administration, documents show the vaccine is 94% effective and well-tolerated.
-
Canada joins the United Kingdom and the United States as the first Western countries to provide the vaccine developed by Pfizer and BioNTech, as the coronavirus pandemic rages toward winter.
-
Illinois received 43,000 doses in its first shipment of a COVID-19 vaccine Monday as health officials reported another 103 coronavirus deaths…
-
"I feel like healing is coming," New York critical care nurse Sandra Lindsay said after receiving her shot. "I hope this marks the beginning to the end of a very painful time in our history."
Play Live Radio
Next Up:
0:00
0:00
Available On Air Stations